QS Blueprint for the Future of Energy Storage
QuantumScape’s blueprint for next-generation energy storage: the technology, the milestones, and the manufacturing foundation built to scale.
A critical factor drivers consider when looking at electric vehicles is how quickly they can be charged (we covered other factors in an earlier blog post). Unfortunately, battery experts can use confusing jargon when talking about this concept, so here we’re going to break down key charging concepts and how they translate to the real world.
The C-rate is the unit battery experts use to measure the speed at which a battery is fully charged or discharged. For example, charging at a C-rate of 1C means that the battery is charged from 0-100% in one hour. A C-rate higher than 1C means a faster charge; for example, a 3C rate is three times faster, so a full charge in 20 minutes. Likewise, a lower C-rate means a slower charge: C/5 (or 0.2C) would be five times slower than 1C, amounting to a five-hour charge.
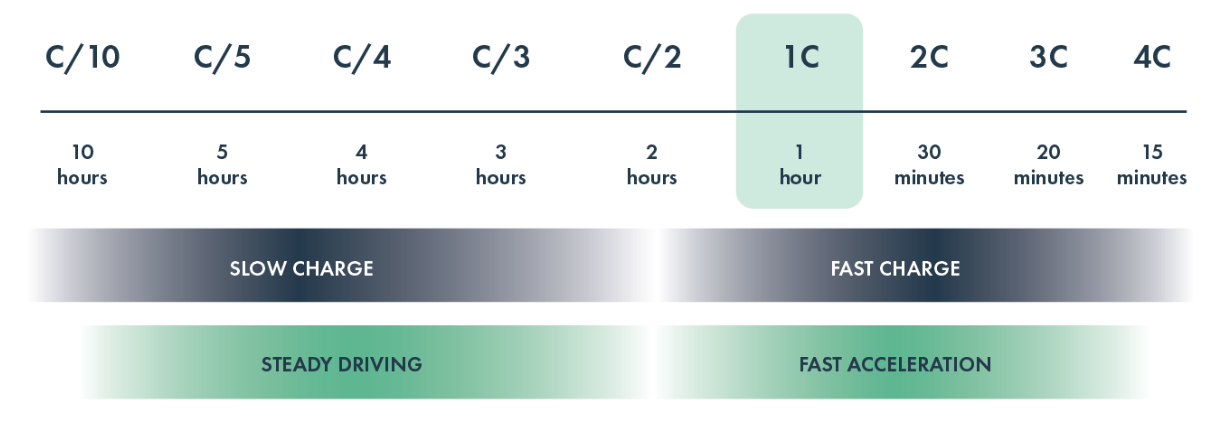
C-rate also is a relative metric. Many important aspects of battery behavior (such as resistance to dendrites) depend on the absolute current density, the amount of electrical current that goes through the battery divided by the area of its layers. For a given C-rate, the current density will be a function of the loading of the cathode, which is closely related to its thickness. Thus, when comparing C-rates, it’s important to ensure the cathode loading is at commercially relevant levels: for EVs, this is typically in the range of 2.5-5 mAh/cm2.
To be a viable alternative, EVs should perform at least as well as their internal-combustion engine (ICE) counterparts: deliver high levels of power during acceleration, operate in all driving conditions, and require minimal time to recharge.
In an ideal world, a battery would perform at extremely high C-rates all the time. However, the higher the C-rate, the more difficult it is for a battery to deliver reliable performance. Higher C-rates increase the rate of degradation in the battery, reducing range and shortening the vehicle’s lifespan. Faster charging can also cause dangerous dendrites to form. These hurt the battery’s lifespan, can lead to cell failure, and in some extreme cases have been known to cause fires. Consequently, EV battery chargers do not sustain charging rates higher than 2C for longer than a few minutes before the charging rate is reduced to avoid causing damage to the battery.
This means that today’s best EV batteries can still only be charged relatively slowly compared to the few minutes it takes to fill a combustion engine gas tank. Of course, EVs have the advantage that they can be recharged at home and overnight, and so fast-charging is usually more desirable when taking trips longer than a single charge (typically around 300 miles) would allow. And the good news is that most batteries regularly charged at slow C-rates will retain their original range for longer than similar batteries repeatedly charged at fast rates.
However, we believe that mass-market EV adoption will require a more comparable experience ICEs. To achieve this, battery technology must be improved to enable a car to charge at substantially faster rates so that drivers can “fill” their batteries in minutes instead of hours. This is a key reason why certain next-generation battery technology is so important.
If current battery technology risks dendrite formation if charged too quickly, are next-generation battery technologies going to solve this problem? Unfortunately, it has been found that not every new battery technology is capable of delivering improvements to fast-charging performance. For example, in our blog post on sulfides, we explained why we believe that many of the newly announced solid-state lithium-metal batteries based on sulfide electrolytes are unlikely to deliver improvements in charging performance over conventional lithium-ion batteries. Similarly, due to the twin issues of dendrite formation and resistance growth, we believe most liquid electrolyte-based lithium-metal batteries are also unlikely to be able to deliver fast charge performance. Though they appear to successfully charge at relatively slow rates, such as C/5, we believe these fundamental limitations to many of the sulfide- and liquid-based technologies are likely to put super-fast charging speeds out of reach.
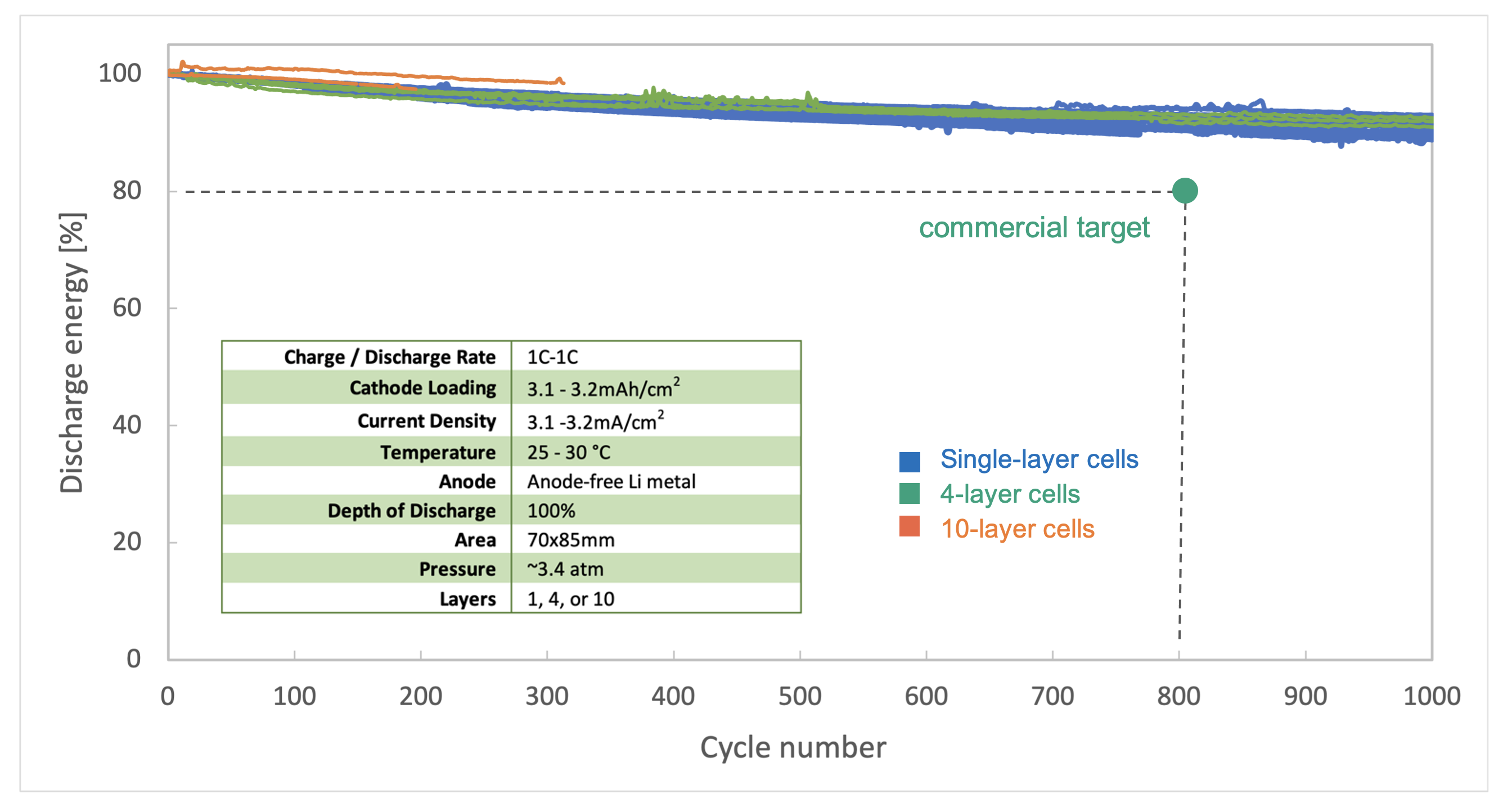
In contrast, our ceramic solid-electrolyte separator enables the use of a lithium-metal anode in its fully charged state. This lithium-metal anode replaces the graphite anode, one of the key bottlenecks to fast charging in current EV batteries. The test results of our batteries using our solid-state lithium-metal anodes show better than 80% energy retention after 800 charging cycles with repeated 1C rates of charge and discharge, the equivalent of over 240,000 miles for a car with a 300-mile range. In short, charging our lithium-metal batteries at a relatively high 1C rate does not cause a dramatic drop in range over the battery’s lifespan.
We believe the data we have shown demonstrates that our fundamental technology, when scaled to a size that is useful for EVs, can deliver the fast-charging speeds that make EVs competitive with ICE vehicles. Our ultimate goal is to make a battery for EVs that can recharge from a low state of charge to 80% in 15 minutes, empowering drivers to switch to zero-emissions cars without having to tolerate the inconvenience of slower recharging currently associated with today’s EVs. We recognize that to make an impact in the real world, we need batteries that don’t compromise on power, charge time, or battery life, and that’s precisely why we design and test our batteries using C-rates that are more reflective of real-world conditions.
Many next-generation lithium-metal battery technologies replace the conventional graphite anode with a lithium-metal anode, which allows the battery to store a greater amount of energy in the same volume. But dendrite formation in lithium-metal batteries is a key reason such batteries have not yet been commercialized at scale.
Dendrites are root-like structures of pure lithium formed within a separator that begin at the anode and grow toward the cathode while the cell is charging. They tear apart the cell from the inside as they grow. When a dendrite reaches all the way to the cathode, they cause the battery to short-circuit and fail. Dendrites are also known to form in legacy lithium-ion battery cells, which have caused fires and even explosions.
It has been observed that for lithium-ion and lithium-metal batteries, higher rates of charge increase the likelihood of dendrite formation exponentially. However, lowering the charge rate in a new battery technology means limiting its ability to be a viable alternative to ICEs or conventional battery-based EVs.
During the battery development process, scientists and experts in a laboratory can control the conditions under which their batteries are tested, allowing them to gain insights into fundamental behaviors. However, such lab tests aren’t standardized or always straightforward, making it difficult to interpret the findings or compare the results with other technologies. A few things to look out for are:
[1] https://insideevs.com/news/507489/tesla-model3-charging-faster-ccs2/
Forward-Looking Statements
This publication contains forward-looking statements within the meaning of the federal securities laws . All statements other than statements of historical fact contained in this publication, including statements regarding the future development of QuantumScape’s battery technology, the anticipated benefits and use cases of QuantumScape’s technologies, the performance and safety of its batteries and demonstration in real-world applications, plans and objectives for future operations, partnerships, commercialization and markets, and expected global demand for batteries and the value thereof, are forward-looking statements. When used in this publication, the words “aim,” “anticipate,” “believe,” “blueprint,” “can,” “committed,” “continue,” “could,” “designed to,” “estimate,” “expect,” “future,” “going forward,” “intend,” “may,” “move,” “must,” “offers,” “plan,” “potential,” “predict,” “pro forma,” “project,” “roadmap,” “should,” “tend,” “target,” “will,” “would,” and the negative of such terms and other similar expressions are intended to identify forward-looking statements, although not all forward-looking statements contain such identifying words.
These forward-looking statements are based on management’s current expectations, assumptions, hopes, beliefs, intentions, and strategies regarding future events and are based on currently available information as to the outcome and timing of future events. These forward-looking statements involve significant risks and uncertainties that could cause the actual results to differ materially from the expected results. Many of these factors are outside QuantumScape’s control and are difficult to predict. QuantumScape cautions readers and viewers not to place undue reliance upon any forward-looking statements, which speak only as of the date made. Except as otherwise required by applicable law, QuantumScape disclaims any duty to update any forward-looking statements. Should underlying assumptions prove incorrect, actual results and projections could differ materially from those expressed in any forward-looking statements. Additional information concerning these and other factors that could materially affect QuantumScape’s actual results can be found in QuantumScape’s periodic filings with the SEC. QuantumScape’s SEC filings are available publicly on the SEC’s website at www.sec.gov.

QuantumScape’s blueprint for next-generation energy storage: the technology, the milestones, and the manufacturing foundation built to scale.
Bringing a new technology to market can seem mysterious from the outside. Headlines tout “breakthroughs” that never materialize, leaving people skeptical. And yet, technology does advance steadily forward – faster computers, cheaper renewables, and better EVs arrive every year. So what separates hype from true innovation?

QuantumScape’s blueprint for next-generation energy storage: the technology, the milestones, and the manufacturing foundation built to scale.

Bringing a new technology to market can seem mysterious from the outside. Headlines tout “breakthroughs” that never materialize, leaving people skeptical. And yet, technology does advance steadily forward – faster computers, cheaper renewables, and better EVs arrive every year. So what separates hype from true innovation?
Privacy Policy | Terms of Use
© 2026 QuantumScape Battery, Inc.
1730 Technology Drive, San Jose, CA 95110
info@quantumscape.com

Pamela Fong is QuantumScape’s Chief of Human Resources Operations, leading people strategy and operations, including talent acquisition, organizational development and employee engagement. Prior to joining the company, Ms. Fong served as the Vice President of Global Human Resources at PDF Solutions (NASDAQ: PDFS), a semiconductor SAAS company. Before that, she served in several HR leadership roles at Foxconn Interconnect Technology, Inc., a multinational electronics manufacturer, and NUMMI, an automotive manufacturing joint venture between Toyota and General Motors. Ms. Fong holds a B.S. in Business Administration from U.C. Berkeley and a M.S. in Management from Stanford Graduate School of Business.